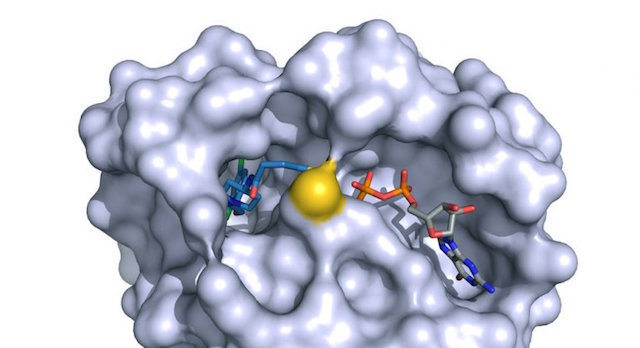
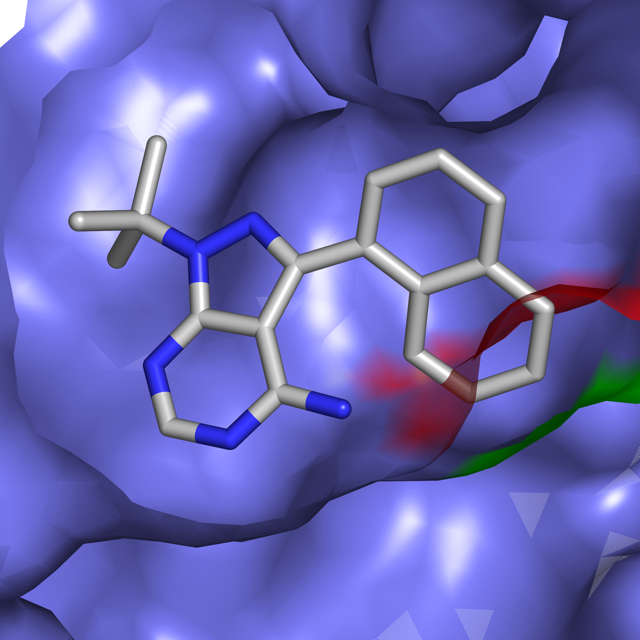

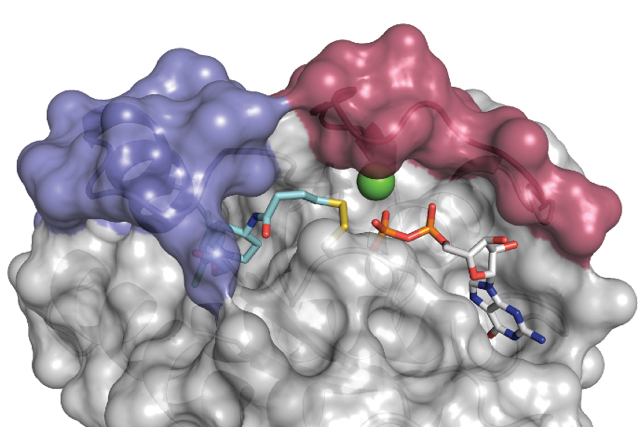
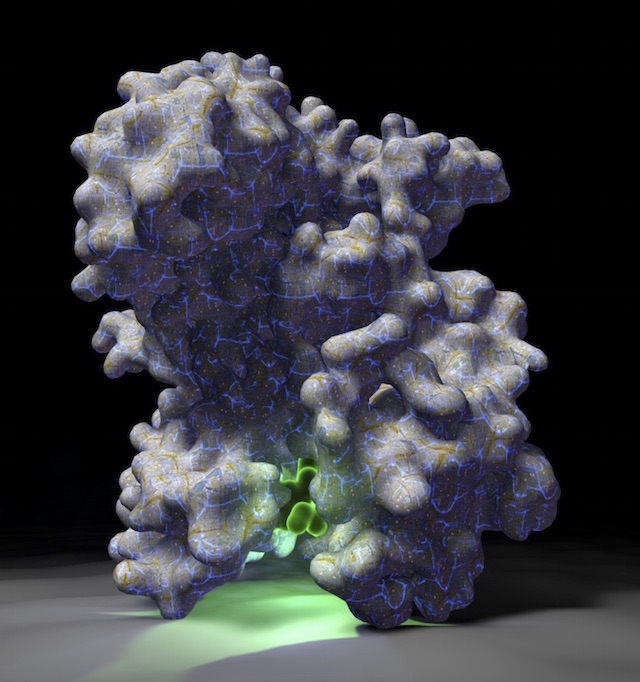
The Shokat lab uses chemical genetics to study and target signaling proteins, with the aim of finding new ways to treat human diseases. Many projects in the lab have focused on kinases, GTPases and RNA helicases, including some of the most important proteins in cancer, such as mTOR, Ras and CDK4. We are also interested in other signaling proteins, such as the dual kinase/GTPase LRRK2, which is relevant in Parkinson's disease, and the translational regulator helicase DDX3.
Chemical genetics combines the timing and dose control of small molecules with the absolute specificity of genetics. Small molecules are invaluable tools for studying proteins, but it can be hard to find one that targets a single member of a family of proteins. We take advantage of the chemical difference that results from a mutation to allow us to target a protein specifically, leaving other similar proteins untouched.
Our lab uses a large variety of techniques. We use synthetic organic chemistry, protein engineering, structural biology, biochemistry and cell-based assays in our projects, learning new tools from each other as we go.